 Because electrons repel each other electrostatically, the most stable arrangement of electron groups (i.e., the one with the lowest energy) is the one that minimizes repulsions. According to this model, valence electrons in the Lewis structure form groups, which may consist of a single bond, a double bond, a triple bond, a lone pair of electrons, or even a single unpaired electron, which in the VSEPR model is counted as a lone pair. We can use the VSEPR model to predict the geometry of most polyatomic molecules and ions by focusing on only the number of electron pairs around the central atom, ignoring all other valence electrons present. 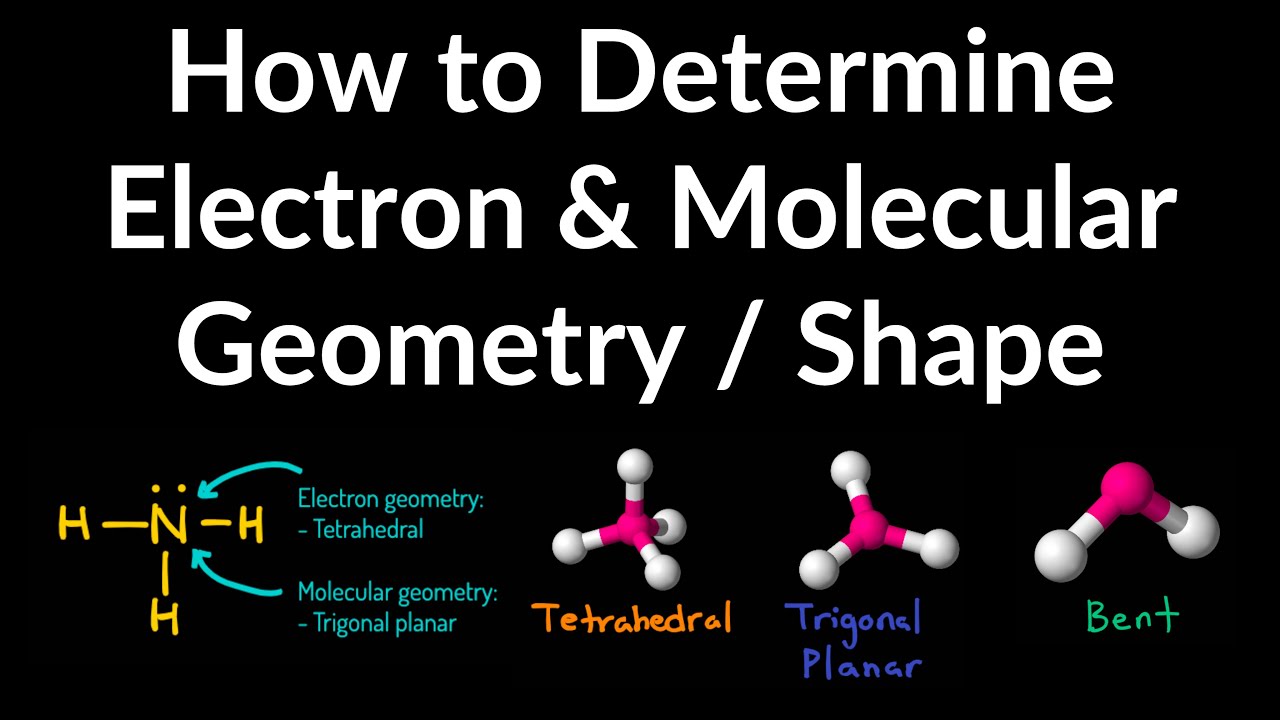
Lewis electron structures predict the number and types of bonds, whereas VSEPR can predict the shapes of many molecules and polyatomic ions. Keep in mind, however, that the VSEPR model, like any model, is a limited representation of reality the model provides no information about bond lengths or the presence of multiple bonds. (pronounced “vesper”), which can be used to predict the shapes of many molecules and polyatomic ions. We continue our discussion of structure and bonding by introducing the valence-shell electron-pair repulsion (VSEPR) model A model used to predict the shapes of many molecules and polyatomic ions, based on the idea that the lowest-energy arrangement for a compound is the one in which its electron pairs (bonding and nonbonding) are as far apart as possible. This approach gives no information about the actual arrangement of atoms in space, however. 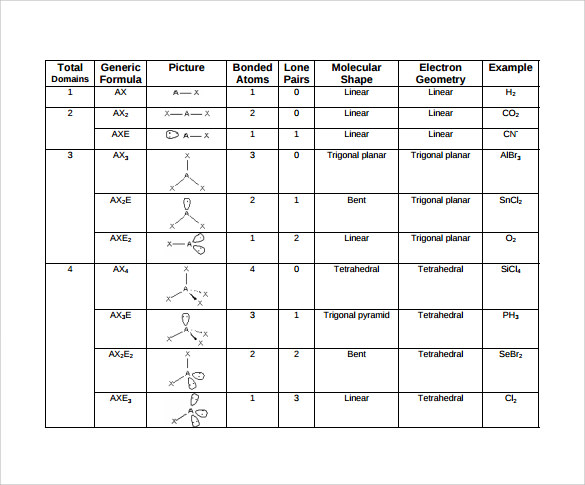
The Lewis electron-pair approach described in Chapter 8 "Ionic versus Covalent Bonding" can be used to predict the number and types of bonds between the atoms in a substance, and it indicates which atoms have lone pairs of electrons. To predict whether a molecule has a dipole moment.To use the VSEPR model to predict molecular geometries.zip file containing this book to use offline, simply click here. 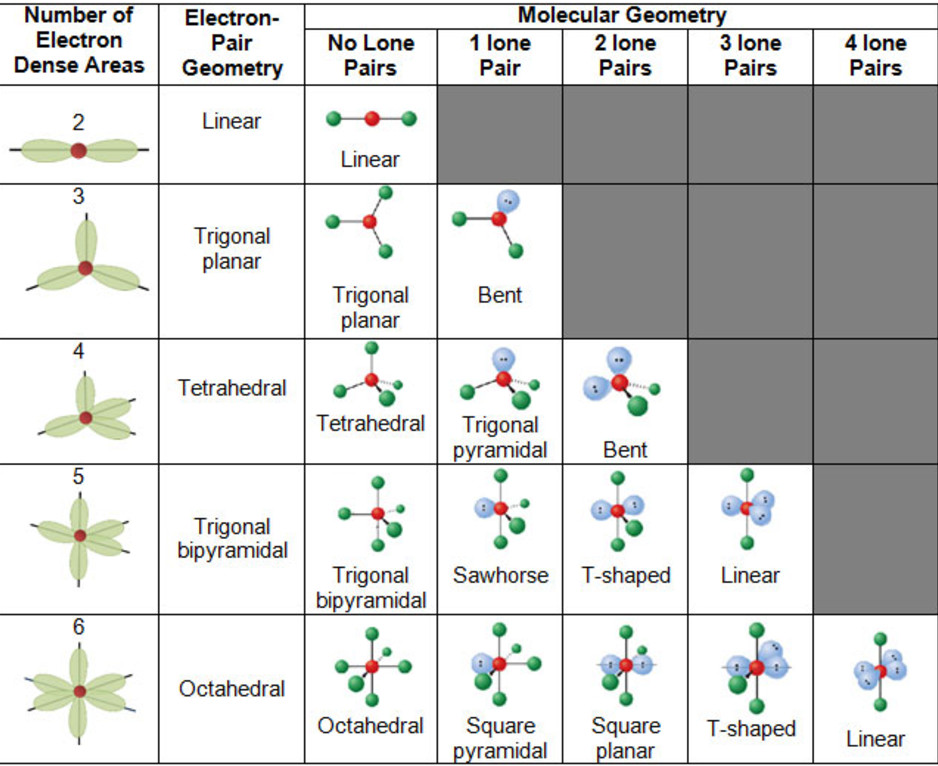
You can browse or download additional books there. More information is available on this project's attribution page.įor more information on the source of this book, or why it is available for free, please see the project's home page. Additionally, per the publisher's request, their name has been removed in some passages. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Normally, the author and publisher would be credited here. This content was accessible as of December 29, 2012, and it was downloaded then by Andy Schmitz in an effort to preserve the availability of this book. See the license for more details, but that basically means you can share this book as long as you credit the author (but see below), don't make money from it, and do make it available to everyone else under the same terms. (g) Six electron groups give an octahedral electron geometry, while four bonding groups and two lone pairs give a square planar molecular geometry.This book is licensed under a Creative Commons by-nc-sa 3.0 license. (f) Six electron groups give an octahedral electron geometry, while five bonding groups and one lone pair give a square pyramidal molecular geometry. 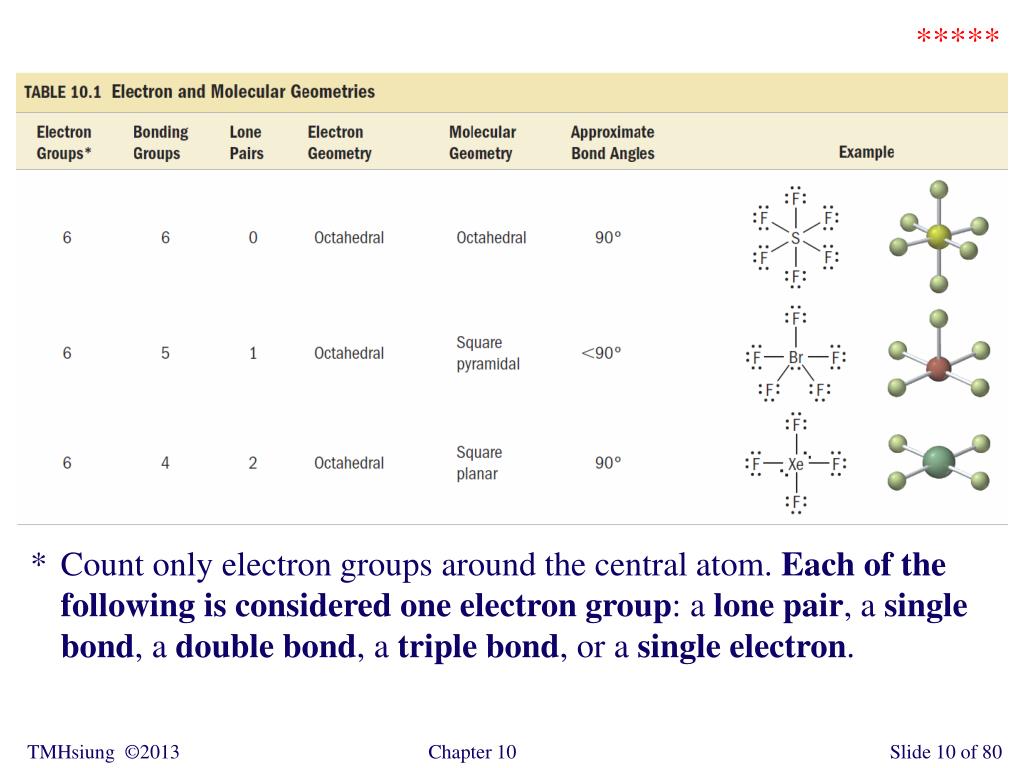
(e) Five electron groups gives a trigonal bipyramidal electron geometry, while two bonding groups and three lone pairs give a linear geometry. (d) Five electron groups give a trigonal bipyramidal electron geometry, while three bonding groups and two lone pairs give a T-shaped molecular geometry. (c) Five electron groups give a trigonal bipyramidal electron geometry, while four bonding groups and one pair give a seesaw molecular geometry. (b) Four electron groups give a tetrahedral electron geometry, while two bonding groups and two lone pairs give a bent molecular geometry. (a) Four electron groups give a tetrahedral electron geometry, while three bonding groups and one lone pair give a trigonal pyramidal molecular geometry. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
